What’s in every science book, every science lab, and every science lover’s brain? It’s the periodic table of elements! The majority of us can identify this fascinating chart when we see it, but many don’t know how to actually read and understand the information displayed on the periodic table.
Most students are introduced to the periodic table at a fairly young age, and it is often taught all the way through high school graduation. So, for all of you high schoolers out there, we’re here to help you better understand this complicated-looking table that willl help you make sense of chemistry.

What is the periodic table of elements?
For starters, let’s take it back to the basics. The periodic table arranges all known chemical elements into a succinct chart for easier identification. It was first professionally published in 1869 by Russian chemist and inventor Dmitri Mendeleev. This original version contained just 63 elements, and that number has grown to 118 today! If a new element is ever discovered, it will be added to the current 118 elements on the table.
I like to think of the periodic table as a chemistry “cheat sheet.” It offers a wealth of knowledge for each of the 118 elements, including:
- The chemical symbol/abbreviation
- The atomic number (the number of protons in the nucleus of the element)
- The atomic weight
- Element type (alkali metals, basic metals, semimetals, etc.)
It’s no wonder we see this fascinating table all over the science world!

How is the periodic table of the elements arranged?
The arrangement of the periodic table is very important in our understanding of it. First, each element is arranged in numerical order of its atomic number. Hydrogen lands in the first spot because it has only one proton per hydrogen atom. The last element, Oganesson, has an atomic number of 118.
Next, we see the elements arranged in periods and groups (columns and rows). Horizontally (columns), elements are organized based on the level of their electron ground state energy. Vertically (rows), elements are arranged in 18 groups, with each group ordered based on electron quantity within the atoms of the elements.
Many versions of the periodic table color code the elements based on what element type they are (metals, nonmetals, noble gases, etc.). This just makes it easier to read and study!
Oh, and don’t forget about the extra block of elements! Directly below the “main” periodic table is a smaller block of elements. There just wasn’t room for these elements in the current table layout, but they’re still important!
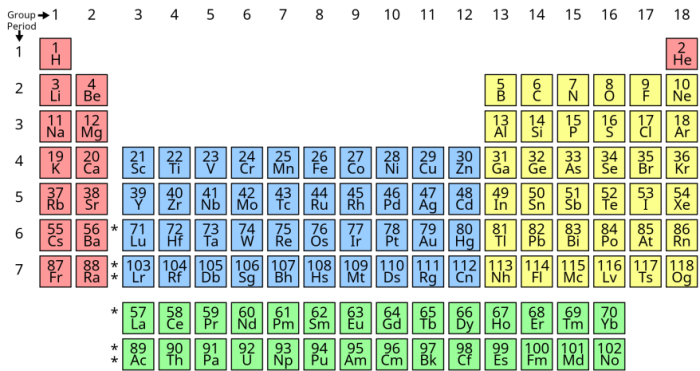
Attribution: User:Double sharp, based on File:Simple Periodic Table Chart-en.svg by User:Offnfopt, CC BY-SA 4.0, via Wikimedia Commons
A Quick Note About the Periodic Table for Kids
Although most younger learners aren’t ready to dive into everything the periodic table entails, it’s never too early to teach the basics. Check out this periodic table scavenger hunt or purchase this colorful, easy-to-read periodic table for kids! Both are awesome ways to start the chemistry conversation with elementary students, and your high schoolers will probably enjoy getting involved too.

Want to learn even more about the periodic table?
As a core part of the science of chemistry, the periodic table is a must-learn for all high school students. That’s why we discuss it in-depth through Experience Chemistry, our online science course for high school. Aside from teaching the basics about the periodic table of elements, our course authors dive into the history, trends, and practical uses surrounding this table.
At Journey Homeschool Academy, we aim to merge high-quality education with fun, engaging content. This includes engaging video lessons, dynamic labs, homework help, and so much more to make the periodic table of elements and chemistry exciting again.
